- Blog
- Uninstall dante virtual soundcard for mac
- West field genius bar appointment san francisco
- Ben 10 games on cartoon network
- Tokyo ghoul unravel japanese lyrics
- Watch free movie 300 rise of an empire
- Adobe illustrator cs3 test
- Typing master 10 amazon
- Coreldraw x6 logo design
- Synfig studio source
- Francotte ampa receptor
- Pdf architect 5 convert review
- To converter pdf a word
- Ez drummer midi
19, 20 While the introduction of the cyclopropyl group in this position was clearly the optimal choice and could thus not be neglected, we decided to also investigate the impact of the insertion of the smallest alkyl radical, namely the methyl group. Substituents inserted on the nitrogen atom at the 4-position were selected according to previously reported structure activity relationships. 19 After alkylation on the 4-nitrogen atom using methyl iodide in the presence of potassium carbonate in acetonitrile and saturation of the double bond in the 2,3-positions by means of sodium borohydride, conversion of the methoxy at the 7-position into a hydroxyl group was achieved with boron tribromide in dichloromethane giving access to 10a. While the intermediate 10b was obtained as previously described, 16 its 4-methylated analogue 10a was prepared via an alternate pathway, starting from 7-methoxy-4 H-1,2,4-benzothiadiazine 1,1-dioxide 7.
FRANCOTTE AMPA RECEPTOR SERIES
The 7-phenoxy-substituted 3,4-dihydro-2 H-1,2,4-benzothiadiazine 1,1-dioxides 11 series were prepared from the corresponding intermediates 10 series through a Chan-Lam coupling reaction with the appropriate phenylboronic acid in presence of copper (II) acetate in dichloromethane.

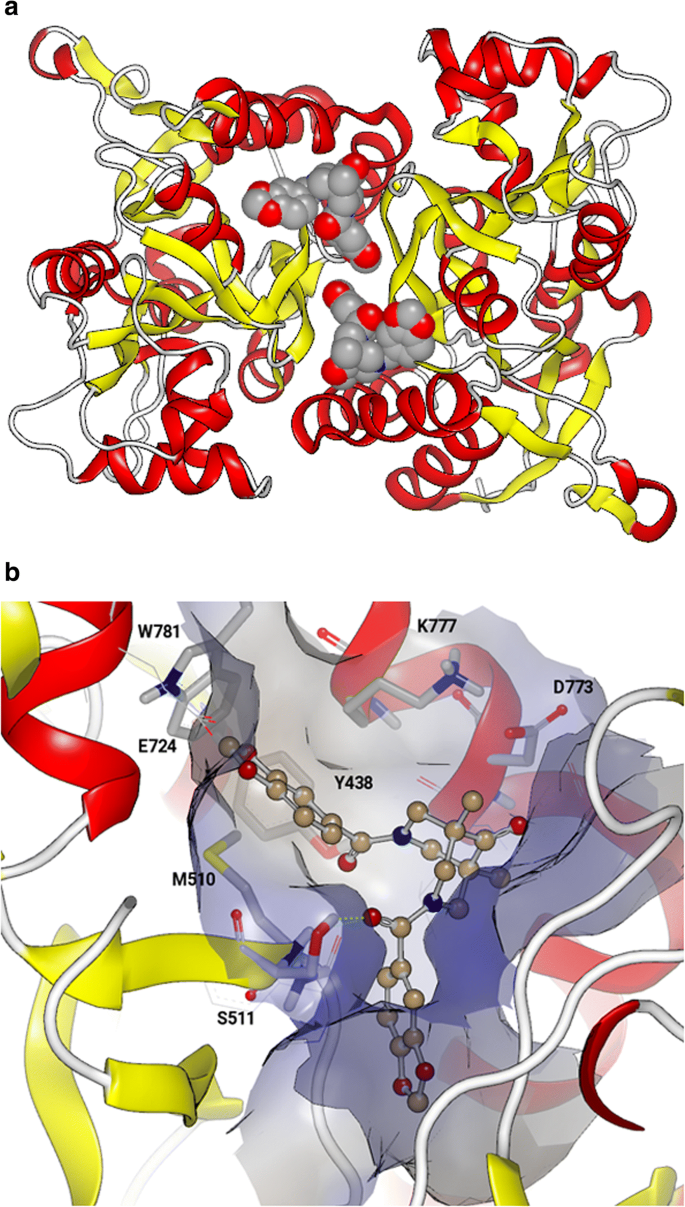
(76%) iv: adequately substituted phenylboronic acid Cu(OAc) 2, molecular sieves, CH 2Cl 2, 40☌, 5 h. 12Īmong the different AMPAR potentiators, 13 3,4-dihydro-2 H-1,2,4-benzothiadiazine 1,1-dioxides constitute probably the most investigated chemical class, as exemplified by recent publications ( Fig. 5, 10 This may be achieved either with one or two potentiator molecules per dimer, thus with two or four potentiator molecules per AMPAR, 11 depending on the occupancy of the A/B/B′/C and C′ subsites which constitute the binding cavity, as previously defined. 9 By their ability to form bridging interactions between LBD dimers, positive allosteric modulators stabilize the receptor in its open state and thus slow deactivation and/or desensitization processes. 8 Each subunit contains an amino-terminal domain involved in subunit assembly (ATD), a clamshell-shaped domain providing the binding pocket for glutamate (LBD), a transmembrane domain forming the permeation channel for cations (TMD) and a cytoplasmic terminal domain (CTD) associated with processes such as receptor trafficking or regulation. While AMPAR agonists are expected to exert excitotoxic effects, AMPAR positive allosteric modulators (so-called ‘AMPApams’ or AMPAR potentiators) seem more promising, as they are able to fine-tune AMPAR signals through their binding at allosteric sites of the receptors and need the presence of endogenous glutamate to be active.ĪMPARs are homo/heterotetrameric complexes assembled with GluA1–4 subunits. 5 This strategy was even found more promising considering their ability to induce the release of neurotrophic factors in vivo, like BDNF. 4 AMPARs are thus considered as an attractive and appropriate target for the discovery of cognitive enhancers and a potential therapeutic tool in the management of Alzheimer’s disease. This idea has been reinforced, taking into account their critical involvement in synaptic plasticity 3 and their involvement in the mechanism of action of nootropic drugs. Amongst these three subtypes, AMPA receptors (AMPARs) have attracted attention, assuming that upregulation of these receptors could lead to cognition enhancement. They have been classified into three subtypes named after their affinity for non-endogenous ligands: N-methyl-D-aspartic acid (NMDA), α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) and kainic acid (KA). 1, 2 iGluRs are recognized to play a crucial role in the fast excitatory synaptic transmission. L-glutamate is well known as the key excitatory neurotransmitter in the central nervous system, acting through activation of metabotropic receptors (mGluRs, coupled to G-protein) and ionotropic receptors (iGluRs, cation channels).
